Beyond the zip: a smarter way to store and access artwork files
Managing packaging assets shouldn't be a guessing game. Yet for many FMCG teams, traditional artwork file management still revolves around bulky zip...
2 min read
 Ekaterina Skalatskaia
:
March 2, 2026
Ekaterina Skalatskaia
:
March 2, 2026

Pharmaceutical packaging artwork is one of the most complex and regulated areas of the packaging world. Every carton, leaflet, label, blister, and insert must meet strict industry standards to protect patient safety — and even the smallest artwork error can lead to costly recalls, regulatory scrutiny, and risks to public health.
As pharma companies accelerate global launches and navigate frequent regulatory updates, manual artwork processes simply can’t keep up. A dedicated artwork management system (AMS) like Cway helps teams streamline workflows, maintain compliance, and eliminate risks that traditional tools cannot solve.
Below, we explore the major roadblocks in pharmaceutical artwork management and how Cway helps companies overcome them with automation, lifecycle visibility, and traceability.
Pharma artwork workflows are inherently complex. They require coordination across:
regulatory teams
quality teams
external agencies
manufacturing sites
packaging development
supply chain partners
Without a centralized system, these workflows become fragmented and error-prone.
Common issues include:
Teams rely on emails, spreadsheets, shared drives, or manual folder structures — none of which offer real traceability.
Manual proofreading, copy updates, version checks, and routing introduce human error and delay approval cycles.
Delays in artwork approval can directly impact regulatory submissions, production dates, and product availability.
Packaging artwork must adhere to FDA, EMA, MHRA, and other regional guidelines — and every change must be traceable.
Cway solves this by providing a single, audit-ready workflow that ensures every stakeholder sees the right file, at the right time, with complete version accuracy.
Pharma artwork is frequently impacted by:
updated ingredient lists
label claims
safety warnings
excipient disclosures
dosage updates
regional market requirements
Each change must be tracked meticulously — often with a corresponding change control number.
Without integration and automation, this becomes a costly challenge.
missed regulatory deadlines
mismatched or outdated content
non-compliant artwork sent to production
increased risk of recalls
unclear ownership and change history
Pharma regulations are time-sensitive — most changes must be implemented within 3 to 6 months, and multiple deadlines must be tracked:
Regulatory Required Date
Effective Date
First Production Date
Missing these dates can disrupt supply, trigger warnings from authorities, or force costly rework.
Tracking these milestones manually is not sustainable.
Cway automates deadline tracking, sends reminders, and ensures all teams stay aligned, reducing the risk of non-compliance and launch delays.
Now that the core challenges are clear, it’s evident that pharmaceutical companies require a dedicated system designed to support compliance-heavy artwork workflows.
Cway empowers pharma teams by providing:
Accurate version tracking is essential for preventing mix-ups and proving compliance.
Cway provides:
automatic version control
full audit trails
access to historical versions
role-based access
Teams always work with the correct version — never outdated files.
Cway helps teams manage submissions and commercial artwork in one place, so everyone can work together smoothly from concept to final artwork.
Manual artwork processes slow down launches and introduce unnecessary risk.
Cway accelerates timelines by:
reminders & notifications
This significantly reduces cycle time and helps pharma companies deliver critical products to market faster — without compromising safety or accuracy.
In the highly regulated world of pharmaceutical packaging, accuracy and compliance are non-negotiable. Manual artwork processes cannot provide the control or traceability required at scale.
Cway gives pharma companies a modern, automated artwork management system that:
enhances compliance
reduces human error
accelerates approvals
ensures traceability
supports complex multi-market workflows
improves speed-to-market
By centralizing the entire artwork lifecycle, pharma teams can confidently navigate regulatory demands and ensure that life-saving medicines reach patients safely and on time.
Explore Cway today and discover how it transforms your pharma artwork workflow.
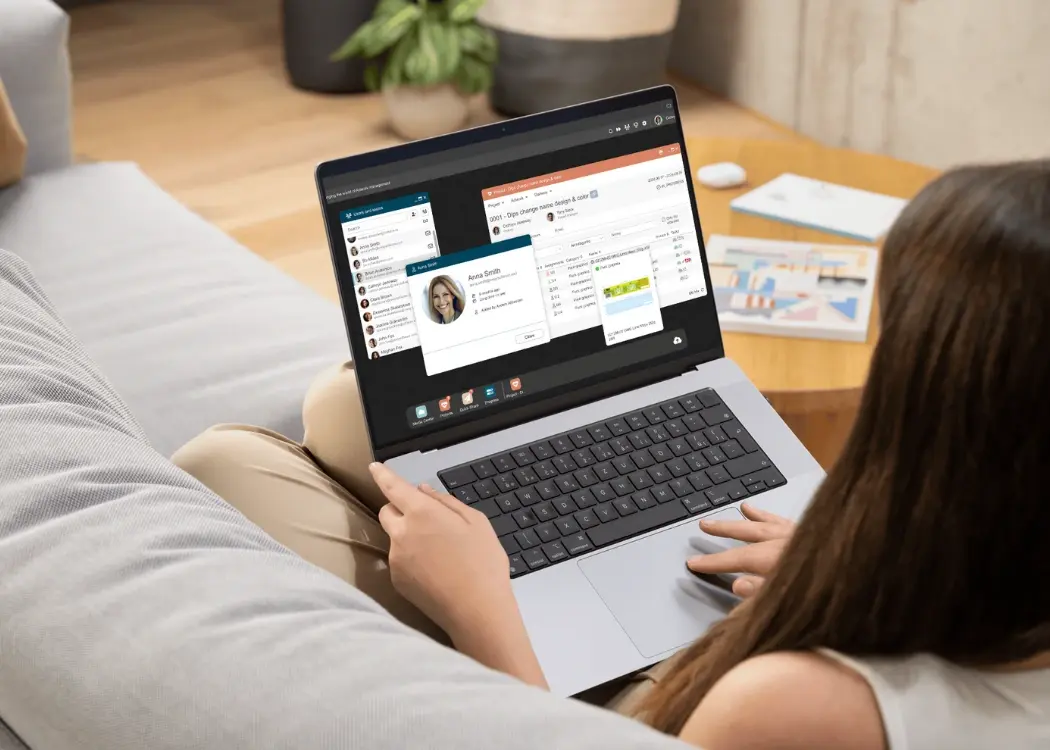
Managing packaging assets shouldn't be a guessing game. Yet for many FMCG teams, traditional artwork file management still revolves around bulky zip...

In this article, we’ll explore what labeling management software is, why it matters, and how it can transform your packaging workflows. Whether...

In this article, we’ll explore how product information management (PIM) plays a critical role in streamlining packaging workflows. From reducing...